Lab 6. Agglutination
When antibodies are mixed with their corresponding antigens on the surface of large, easily sedimented particles such as animal cells, erythrocytes, or bacteria, the antibodies cross-link the particles, forming visible clumps. This reaction is termed agglutination. Agglutination is a serological reaction and is very similar to the precipitation reaction we learnt last week. Both reactions are highly specific because they depend on the specific antibody and antigen pair. The main difference between these two reactions is the size of antigens. For precipitation, antigens are soluble molecules, and for agglutination, antigens are large, easily sedimented particles. As you will see from this lab exercise, agglutination is more sensitive than precipitation reaction because it takes a lot of more soluble antigens and antibody molecules to form a visible precipitation. To make the detection of soluble antigen and antibody reaction more sensitive, a precipitation reaction can be transformed into an agglutination reaction by attaching soluble antigens to large, inert carriers, such as erythrocytes or latex beads.
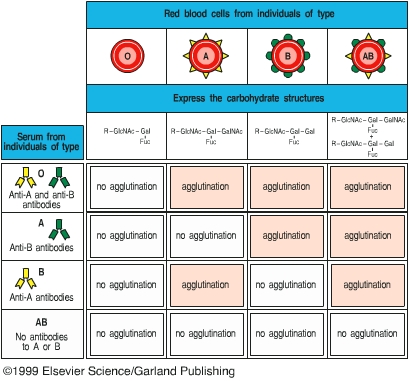
Agglutination reactions have many applications in clinical medicine. Agglutination reactions can be used to type blood cells for transfusion, to identify bacterial cultures, and to detect the presence and relative amount of specific antibody in a patient’s serum. Agglutination has been commonly used to determine whether a patient had or has a bacterial infection. For example, if a patient is suspected of having typhoid fever, the patient’s serum is mixed with a culture of Salmonella typhi. If an agglutination reaction occurs, shown as clumping of the bacteria, the patient either had or has an S. typhi infection. Since certain antibodies can persist in a patient’s blood for years after the patent has recovered from the infection, a positive reaction does not mean that the patient currently has the infection. To determine whether a patient is currently suffering from typhoid fever, the amount or titer of the antibody will be determined at the onset of illness and two weeks later. If the titer of antibody in the patient’s serum has increased at least four-fold between the two tests, the patient is currently fighting off the infection, and the pathogen causing the illness is confirmed.
In this lab exercise, you will learn two different methods of employing agglutination reactions, rapid slide agglutination and microtiter test. These two tests are valuable methods commonly used in clinical laboratories. Applications of agglutination include A-B-O blood typing tests and rapid bacterial identification. The microtiter test is used to quantify the amount of antibody in patient’s blood.